|
|
Post by szczepanek on Jan 18, 2017 1:48:41 GMT -8
Hi rocket scientists! My first post here, my fingers getting a little shaky.  I’ve only learned about rocket stoves five months ago, when looking for a better solution to heat up the old house I live in. I’ve gotten fairly deep into it though, to the point that I got asked to translate the best part of Peter Berg’s batchrocket.eu into Polish. Very pleasant work. Hi Peter!  I’d like to bounce my idea for a rocket hot bath off you. I wanted something a little sexier than a heat exchanger stacked on the stove and connected to the bath via pipes. Also, I recalled my last winter dip in the “chan”, a type of bath common in the highlands of Western Ukraine (see spravka.ua/company/photo/4f6ce01fab4bc.jpg). The idea is to employ a J to spit hot gases underneath the cast iron bath (ca. 170x70 cm = ca. 67”x28”). The exhaust is at the opposite corner of the bath. Under the bath there are fire bricks in order for the bottom of the bath not to get too hot, and for some mass. There are holes in the lower layers of this fire brick structure for the gases to flow through. 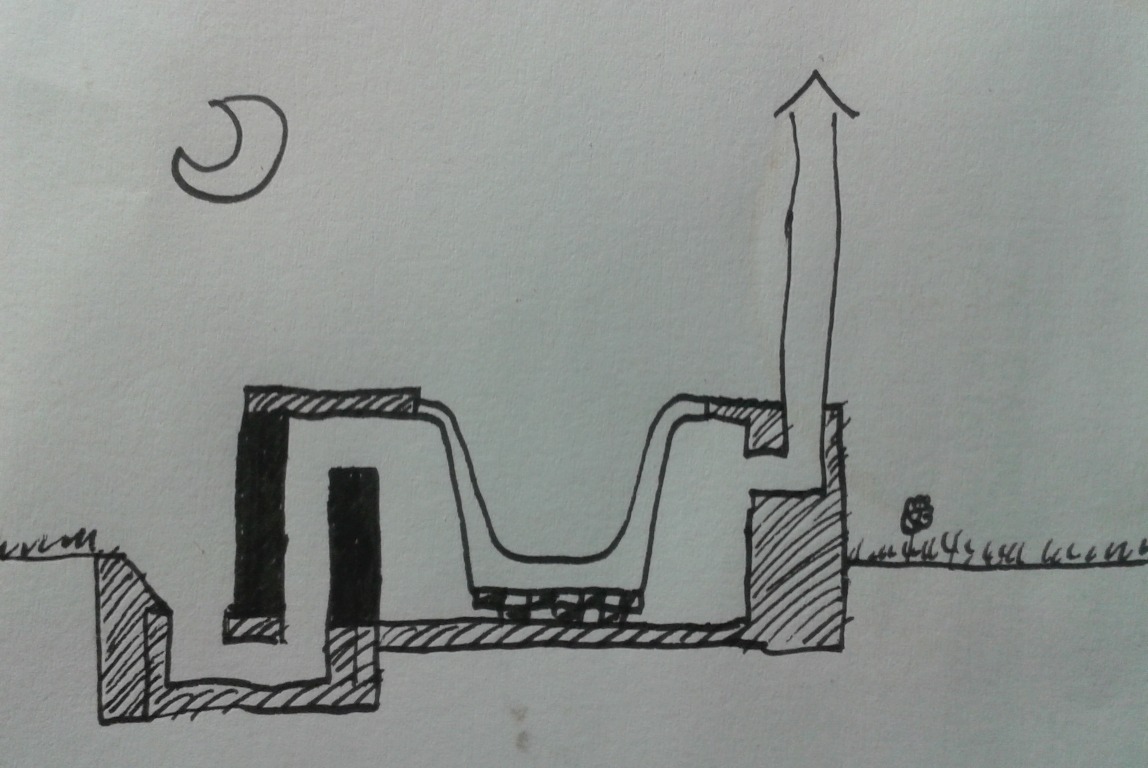 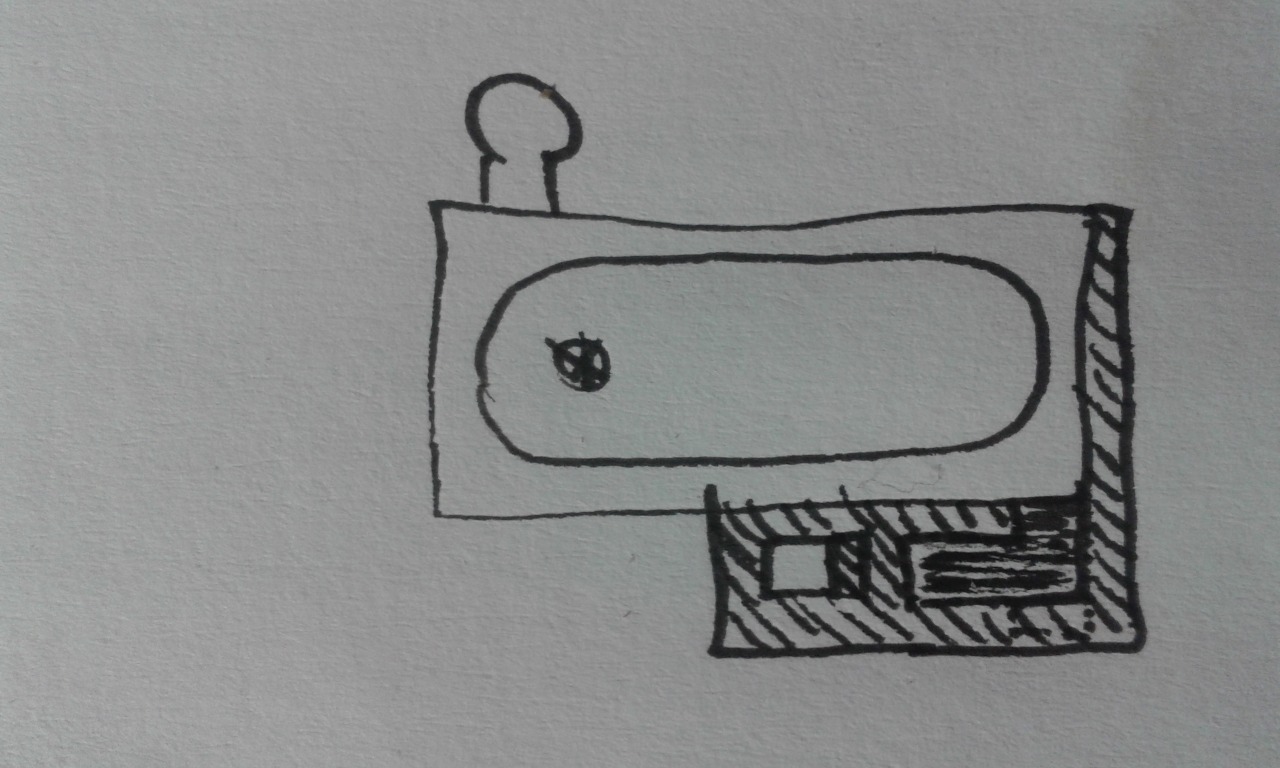 Will it work? Is it OK that the gases make a 90 degree turn and travel horizontally for a moment after leaving the riser and before entering the bell? How do I calculate the system size once I know the ISA (which depends on the bath size)? Is it not going to take hours before the water gets hot? Is the exhaust height right or would you make it lower? Do you know of similar projects? Thank you! |
|
|
|
Post by satamax on Jan 18, 2017 2:40:15 GMT -8
Check the discutions at permies. There's a few who have delved far into rocket baths.
|
|
|
|
Post by peterberg on Jan 18, 2017 4:43:47 GMT -8
Hi Szczepanek, welcome to the boards.
As it is drawn, the rocket side of the bath is heated first and, more importantly, the part above the water level is heated best. This isn't a safe construction, that part of the bath is getting scorching hot. The other side won't be heated that much, the hotter gases are going to the top immediatly and vented to the great outdoors.
Start with bringing the bath higher up in such a way that the part above the water surface isn't heated. What I would recommend: make a lot of space all around the bath so the hot gases can go to the other side unhindered and shift the exhaust opening to as low as possible. Think of the whole thing as a bell: the hot gases tend to rise, thereby displacing the cooler gases to the exhaust, the lowest part of the bell. The bath is nothing more than a big recess in the top of the bell, so to speak.
|
|
|
|
Post by szczepanek on Jan 25, 2017 14:30:45 GMT -8
Thank you for your replies! I followed your suggestions, Peter, when drawing the 3D model. The bath is now a bit higher up and its upper part remains out of the bell. The gases can flow around or underneath the bath to reach the other, cooler side. Is it what you meant? 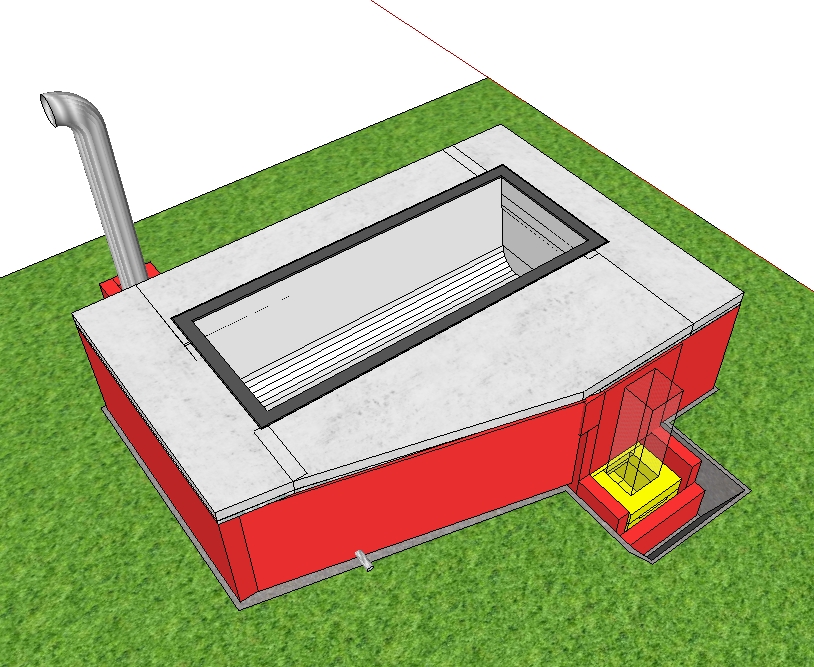 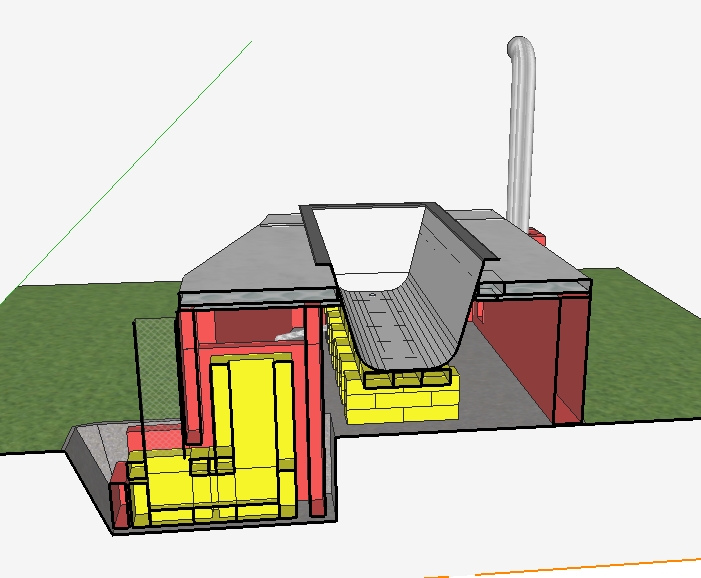 SKP file: tiny.pl/gdswz What I didn’t put on the pictures is the insulation of the external walls - initially I thought 12 cm of standard brick is not bad, but I imagine the thing would be much more efficient if I put ceramic fibre blanket on the internal side of all the external walls (I just wonder how much it’s going to cost for that pretty big surface). I’ve read a few threads on permies and now I’m even more concerned with how much time and wood it will take to make my bath hot, especially when there’s less than 10°C outside and no sun to pre-heat the hose. How would you approach calculating these things? |
|
|
|
Post by peterberg on Jan 26, 2017 1:17:34 GMT -8
Looks much better now. One snag though: the exhaust pipe looks much smaller than the J-tube. In order to avoid restrictions, this pipe should be the same as system size. That insulation should be all around the walls and the ceiling of the bath bell. Especially the area above the riser, that will be the hottest spot of the whole construction.
I can't tell you how long it will take before the water will get warm and how much fuel you need, sorry. Someone else might have paid more attention in school than I did.
|
|
|
|
Post by matthewwalker on Jan 26, 2017 8:10:46 GMT -8
I'm going to guess a long time. All day.
|
|
|
|
Post by scottiniowa on Feb 3, 2017 6:26:52 GMT -8
About how long and how much wood: This is just using known or perhaps better said, estimated math.
if you use an average of 5000 btu's per pound of wood (yes, some better/some worse) a fairly close figure. And use 1 btu to raise one pound of water 1 degree- (universal, calculation I think) And have 30 gal of water in your tub,
(a lot, but a large hot bath, looks to be the goal)
So, 30 gal x 8 lb is 240 lbs of water-
240lbs come in at 50 degrees and you want to get to 104 degrees or 54 degree rise x 240 lbs water or 12960 btu's required.
Now you divide that by your btu's of wood, and you can see 2.5 to 2.6 pounds of wood. This would be NO other heat loss, 100% efficient collection, and everything perfect. Which we all know is not true.
So if we go back to our best hope of efficiency of 70% (2.5/.7) we get about 3.5# of wood. To do the job for 1 minute.
Now the hard part, calculations of heat loss during bath, heat loss to maintain, and start up time loss, all come to play.. And of course, the amount of wood left in stove after bath is done, All come to play.
I would never suggest that this could be done for a mere 3.5 # of wood, but would say this is getting you in the right direction. AS always, I don't mind a bit to be corrected, countered or simply called wrong. Please do tell if there is a far better way to calculate.
|
|
|
|
Post by Vortex on Feb 3, 2017 11:03:37 GMT -8
My neighbour has a hot-tub with a commercial japanese wood fired heater. It takes a whole day to get it up to temperature from cold, and then he'll use a full bale of peat brickettes a day to keep it hot. He won't use wood because he says it doesn't get hot enough and you have to feed the fire to often, whereas the peat brickettes burn hot and slow.  |
|
|
|
Post by szczepanek on Aug 30, 2018 2:20:27 GMT -8
Hi again, As you can easily guess, this design hasn't been realised (yet). It's thanks to you making me imagine the amount of fuel it would take  I pondered about it for some more time (at the time) and, not feeling the then SHELVED it for good (what a cute addition to my English BTW!). Now, after some more stovemaking experience, I'm after building a proper heater at home, which I'm just about to describe in another post. Thank you for your attention here! |
|
|
|
Post by Orange on Sept 1, 2018 12:29:31 GMT -8
I like your original pic:
so with a rocket built underground and some side insulation it should heat up faster and use much less wood. Also the bottom part heats the most which is ok.
|
|
mudder
Junior Member
 
Posts: 50 
|
Post by mudder on Sept 3, 2018 15:55:48 GMT -8
szczepanek i think you could be more efficient with the tub over the riser, instead of over the bell. or maybe somehow do both? or do like the picture and just make a fire under the thing and make a people pot.  hot tubs are cool projects no matter how you do it tho. i have sauna thoughts in my brain that keeps popping up. |
|
|
|
Post by kiwinutter on Sept 3, 2018 16:48:29 GMT -8
Hi ive built one and she's pretty damn good now 4 burns in a 4 inch peterberg batch and she's hot.
Dont know how to post photo's tho.
|
|
|
|
Post by Vortex on Sept 3, 2018 23:22:39 GMT -8
The forums picture hosting is full so you have to upload your pictures to another site and link to them from here. imgur.com/ seems to be the most popular and reliable site for free picture hosting that allows 3rd party linking. Don't use photobucket, it's a scam now.
|
|
mudder
Junior Member
 
Posts: 50 
|
Post by mudder on Sept 5, 2018 6:26:10 GMT -8
you ever think about using thermal syphoning in conjunction with the plan u have? mite speed up the heating?
|
|
|
|
Post by szczepanek on Sept 7, 2019 22:44:28 GMT -8
kiwinutter , please do send the pics of that! I guess you've got much more experience with it by now? mudder , I have thought about it but I originally wanted to have it simpler than that. 3 years after, I'll definitely reconsider it!
|
|